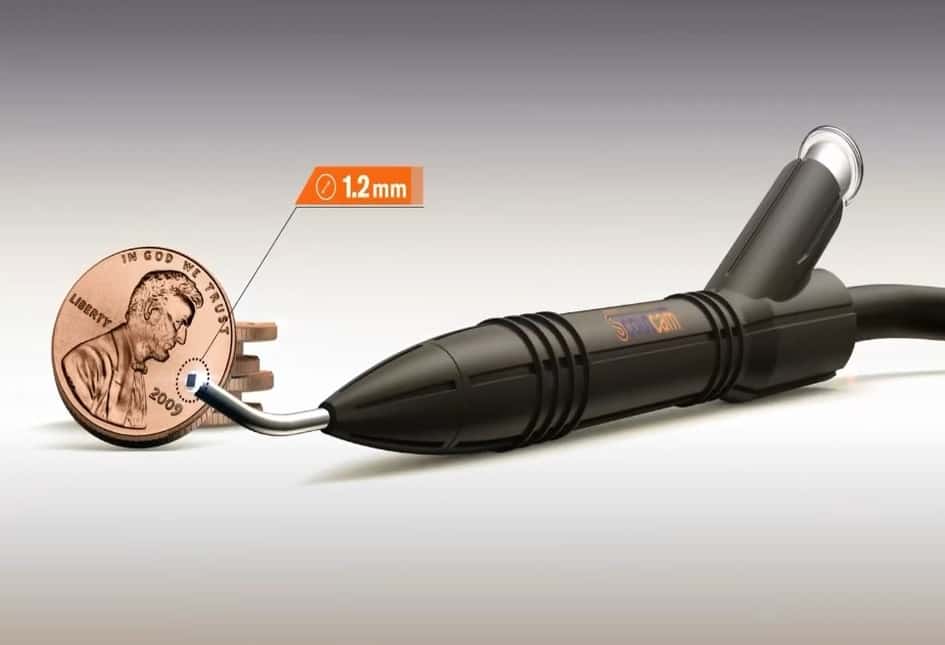
Israeli firm ScoutCam was spun out of Medigus — a medical device company that specialises in minimally-invasive endosurgical tools — in 2019
From diagnostic procedures to minimally-invasive surgeries, miniature cameras have a multitude of uses in the medical sector. ScoutCam currently makes the world’s smallest single-use camera — a device that has been deployed across several industries during the Israeli company’s 20-year history. Jamie Bell speaks to ScoutCam CEO Yaron Silberman to find out more.
The debate over disposable, single-use medical devices versus reusable equipment is by no means a new one, with the pros and cons of both compared for decades.
The major selling point of reusable devices is that they last longer, and can be considered more environmentally friendly because they produce less biomedical waste.
Disposable devices, on the other hand, are looked upon as safer — because they don’t carry the risk of infection through use on multiple patients — and increasingly cheap to make and purchase.
In recent years, the problem of cross-contamination has become more of a priority for healthcare providers — particularly when it comes to endoscopes, laparoscopes, and other surgical instruments that are inserted into the body.
As recently as August 2019, the US Food and Drug Administration (FDA) recommended manufacturers transition from reusable duodenoscopes, which are used to diagnose and treat problems in the pancreas and bile ducts, to models with disposable components, in an effort to prevent the risk of infection among patients.
While this shift may have proved challenging for some companies, Israeli firm ScoutCam found itself ahead of the curve when the FDA made this announcement.
The company has close to 20 years’ experience in making single-use, disposable cameras with applications in several areas — with medical devices one of the most notable.
ScoutCam CEO Yaron Silberman says the firm’s mission is “to enable direct visualisation in difficult-to-reach places” and, in doing so, allow organisations to improve the products they make, and improve the procedures these products are used in.
“We have 20 years of experience in doing this,” says Silberman. “And we’re always working on seemingly impossible missions.
“Our CTO keeps saying: ‘Nobody comes to us with easy problems — because if they were easy these companies would solve them themselves’.
“They come to us with the most problematic situations — either mechanically, electronically, or environmentally — and we have the reassurance that we have been doing this successfully for 20 years.
“We’re not saying that other engineers can’t do it too, but we can save time in doing so, and save costs as a result, because we have crossed some bridges that others still haven’t.”
History of ScoutCam
ScoutCam was originally spun out of an Israeli medical device company called Medigus, which specialises in minimally-invasive endoscopic tools.
In 2010, Medigus had launched what was, at the time, the smallest video camera in the world with a diameter of 1.2mm.
The product — which Silberman describes as “truly revolutionary”— was given the brand name ‘ScoutCam’.
When Medigus decided to rethink its overall focus in light of decreasing share prices, the same title was given to the spin-off company it entrusted with taking the miniature camera device forward.

ScoutCam was officially launched on 1 March 2019, with many of Medigus’ team — including Silberman, who joined in 2011 — also moving across to the newly-formed company.
Today, ScoutCam undertakes projects with companies working in many subsectors. The most notable of these to date was NASA, which used one of its miniature HD cameras during a robotic refuelling mission in 2018.
The Israeli firm does, however, often find its products being used in medical devices — partly because many of the staff members who transferred from Medigus have a background in the broader healthcare industry, but also because there are many real-world advantages to making precise endoscopic tools as small as possible.
What does ScoutCam do?
Its flagship product, the CMOS (complementary metal-oxide-semiconductor) micro ScoutCam 1.2 is, to the best of the company’s knowledge, the smallest device of its kind in the world — with an outer diameter of about 1mm.
The camera also boasts a minimum focus distance of about 2mm, allowing it to produce “superior image quality”, and its durability has been tested under severe environmental conditions including extreme temperatures, vibrations, and radiation.
The vast majority of ScoutCam’s work in the medical device field can be divided into two main categories: minimally-invasive surgeries, and diagnostic endoscopy.
One of the most common types of surgery the company’s cameras are used in is orthopaedics — which includes everything from bone grafts and soft tissue repairs, to joint replacements and fixing broken bones.
Many of these procedures have now moved away from open surgeries, and are instead performed using minimally-invasive laparoscopy, whereby small incisions are made in the abdomen, or endoscopy, in which a catheter is inserted into the patient’s body.
In diagnostics meanwhile, Silberman says ScoutCam’s camera is most commonly used in gastrointestinal-related procedures — which involve investigating the stomach and intestines — but has also seen applications in areas such as cardiology and urology.
As well as these traditional uses of its miniature cameras, the company has recently started to work with medical device manufacturers to investigate organs that were previously not considered relevant to endoscopy.
ScoutCam’s endeavours in this space have seen its technologies used in “very sensitive” areas like the eyeball or the inner ear.
As Silberman says, this process of ‘microendoscopy’ is only possible because the cameras involved have a similar width to a needle, meaning the bodily tissue that needs to be penetrated can heal by itself afterwards.
The future of single-use devices
While the cost and additional safety of using disposable medical devices has seen them become increasingly popular within the industry, the opposite trend can be observed in many other areas.
Water bottles, shopping bags, and straws are among the everyday, single-use items that have come under scrutiny for their impact on the environment, and are subsequently being replaced with reusable alternatives — while rechargeable batteries and lightbulbs are also a relatively recent phenomenon, introduced to help reduce waste.
With the desire for cheaper, safer disposable options beginning to clash with these calls for reusable, but also more expensive, items, Silberman anticipates that the former is likely to eventually come out on top — in the medical sector, at least.
“There is a conflict between environmental factors, and health and safety factors — in medical devices, they are really going in opposite directions,” he says.
“My gut feeling is that, in this area, the safety element will eventually win, and more devices will become single-use.
“I think that, in order to fully resolve this conflict, however, people will have to develop more clever ways of disposing of these single-use devices too.”
Another “interesting concept” Silberman is currently observing in medical devices, is growing demand for imaging sensors that are smaller, but also specifically designed for use in medical applications, with properties tailored to the human body.
“Everything that is very new now will be old in two years and obsolete in five — so, from a business perspective, ScoutCam is going to continue to partner with companies and provide the very critical component of visualisation,” Silberman adds.
“In medicine, smaller is better — but there are things we’ve noticed that are being negatively affected when you try to reduce the dimensions of the camera even further.
“The pixels of today’s cameras are getting down to the size of the photon, so we really are reaching the borders of physics.
“But, there are still ways to improve the performance of the cameras while preserving the dimensions, or even to push the dimensions down while maintaining the same level of quality.”
To read the full article on NS Medical Devices
Four months after the coronavirus began to drastically change life in the U.S., there’s finally some good news after Pfizer was awarded $1.95 Billion to produce a vaccine by year’s end. In 2020, one thing is for certain, COVID19 has drastically changed the way we approach medical processes, and even the medical device development industry has undergone a massive transformation.
On one hand the need for innovation across healthcare and for new medical devices to join the market has been increasing; On the other existing device producers had to innovate in other to navigate the ongoing pandemic.
Since March, the healthcare industry has been continuously evolving. Improvements were made where needed. New devices joined the market, and now we are witnessing the development of a vaccine that, if successful, can eventually control the pandemic.
To navigate the impact of the virus many medical device providers and healthcare suppliers had to change their processes of operation to meet isolation guidelines, while still trying to create products needed by healthcare facilities. Additionally, travel restrictions negatively affected the interaction between engineers and quality teams from both sub-suppliers and device manufacturers, leaving companies to innovate to move forward.
Innovative Trends that Emerge
As the medical industry was forced to shift during the pandemic, interesting trends emerged. First, medical teams became society’s heroes, while public attention was focused on their challenging and serious work, especially with the entire healthcare system disrupted.
Second, tremendous financial and medical resources were transferred to meet demand for the devices needed to combat COVID-19, such as ventilators. Other, less relevant devices diminished in priority.
Finally, in-person communication methods were completely disrupted and video took on new emphasis, becoming synonymous with this era. Now, the trust in video technology as a major vehicle for communication is here to stay.
The use of video allows for remote diagnostics and a way to get information from inside the body and presents a new wave of minimally invasive therapeutic procedures. Hence, video technology, and the production and delivery of these products, plays a critical role in a time when only essential surgeries are taking place.
Beyond video, the medical innovation that will play a larger role in the medical world moving forward as we wait for a vaccine, is directly linked to video technology. In addition, the best practices established under strict limitation and constraints might have long-term, positive impacts on the healthcare industry.
ScoutCam develops and manufactures customized visualization solutions.
To read the full article on HealthTech Hotspot
In the modern medicine and according to the biohazard epidemiological principles, there is a clear tendency towards single use (disposable) invasive devices.
The increase in fears of blood-borne pathogens like HIV, HCV or even prions helped ignite the growth of the Single Use Device business model in the medical industry.
It has been applied to sectors from simple syringes to complicated surgical devices. Indeed, the disposable model has many benefits. Disposables eliminate a key point of contamination, improve hygiene and give patients and practitioners great peace of mind.
The FDA, May 2013, renewed its focus on reusable medical devices after receiving reports that patients were exposed to infection when the instruments weren’t properly cleaned, hence the FDA is taking steps to reduce the risk of infection from reprocessed reusable devices.
One of the best solutions to prevent patient contamination or biohazard exposure is the single use invasive medical devices.
To eliminate the risk for patient contamination and to improve patient hygiene, ScoutCam chose to apply a single-use camera in its invasive devices.
Endoscopes are evolving to minimize infections and the impact of procedures. How can technology help?
In order to fully understand how technology could transform endoscopy and why the space needs innovation, we must start from the beginning of the use of endoscopes.
The History of Endoscopes
Philipp Bozzini of Mainz, Germany, created in 1806 the first internally illuminated device used to inspect the interior of the human body, according to archives from the American College of Surgeons. The device was made out of a tube, with various attachments, that could be safely inserted into a body cavity, it was explained. A candle and angled mirrors inside the device carried light into a human cavity, such as the larynx, the female urethra, and the rectum, allowing the physician to see inside.
The primary goal of an endoscope is to carry light from outside the body into a body cavity. The reflected light, which offers the image of the patient’s body, is then carried back up to the physician’s eyepiece, or into a camera that can display the image on a TV monitor or and optical channel.
Today, “most modern endoscopes aren’t limited to [just] piping light in and out of a patient’s body,” according to Explain that Stuff. They can also be used to carry out small surgical operations and other minor, medical procedures through a separate, working channel built into the endoscope.
Typically, a third and separate channel built into an endoscope allows water irrigation at the distal tip of the endoscope to improve visualization.
Over time, as endoscopes continued to evolve, improvements were made in light sources and optical lenses. Cameras were placed at the distal tip of the endoscope, with the image carried along an electrical cable to outside the body in some endoscopes, replacing the need for fiber-optics, and allowing for smaller and more flexible endoscopes.
Developers can already envision how they see the endoscope transforming—they just need the tools to get there.
What’s Next for Endoscopes
As the Cheshire Cat in Alice in Wonderland told Alice (in so many words), first tell me where you want to go so that I may tell you which road to take.
Ideally, endoscopes should become cheaper, smaller, and more flexible, with larger working channels, and ideally be single-use endoscopes to prevent cross-contamination that can occur with multiple-use endoscopes.
These goals may be achieved by:
- Using inexpensive high-resolution/definition single-use micro cameras.
- Unifying the optical channel with the irrigation channel by allowing water to flow in and around the optical components.
Through such technology, endoscopes could either become smaller and more flexible or alternatively allow enlargement of the working channel. This would allow for more surgical procedures to be carried out in a minimally invasive way, reducing the number of open surgery procedures performed today.
Above: Micro-CMOS sensor cameras startfrom 1.2mm and go up to 3mm.
The importance of single-use endoscopes was recently recognized by the U.S. FDA. The agency recently explained that it “believes the best solution to reducing the risk of disease transmission by duodenoscopes is through innovative device designs that make reprocessing easier, more effective, or unnecessary. For example, duodenoscopes that incorporate disposable components can facilitate cleaning, reduce contamination and reduce disease transmission following reprocessing. Disposable designs may reduce between-patient duodenoscope contamination by half as compared to reusable, or fixed endcaps.”
For instance, single-use duodenoscopes can be developed for bile duct procedures, and they should be implemented for endoscopies in other body cavities, especially those cavities that are sterile and more susceptible to infections such as the urinary tract, brain, spinal cord, and more.
Additionally, other goals that should be taken into consideration include improved optical systems and use of computerized vision and machine learning to help physicians avoid missing lesions.
As both endoscopes and FDA regulations continue to evolve, the three goals listed above seem to be the most urgent and easily achievable today.
To read the full article on mddionline.com
Since the dawn of modern medicine, doctors have pursed the use of technology to help them see into the human body. A variety of means serve just this purpose: X-rays, CT scans, optical fibers and more, but the greatest breakthrough in the field can be attributed to a unique technology developed by the Israeli company Medigus
The Israeli company Medigus developed the smallest camera in the world, at 1.2 mm diameter, which can be employed in medical procedures, in particular endoscopic diagnoses and treatments. The micro-camera’s miniscule diameter allows doctors to see into tiny organs and systems which were previously inaccessible.
Medigus CEO Dr. Elazar Sonnenschein explains that the only alternative are optical fibers which let doctors film video footage inside the human body although their numerous disadvantages limit their use: “Optical fibers are breakable and therefore can’t access many spots. They’re also very expensive – the smaller the diameter the higher the price,” Sonnenschein explains.
The Medigus camera is used in flexible devices that offer a glance into places in the body that the optical fiber cannot access. In addition, it costs roughly 90% less that optical fibers of the same diameter. The low cost allows for significant savings in the component parts of the systems they comprise, and even allow the device to be disposable.
Single-use cameras bypass expensive sterilization Since the dawn of modern medicine, doctors have pursed the use of technology to help them see into the human body. A variety of means serve just this purpose: X-rays, CT scans, optical fibers and more, but the greatest breakthrough in the field can be attributed to a unique technology developed by the Israeli company Medigus / Lior ilam processes, thus also avoiding the risk of dangerous contamination and infection as well as cutting down on excess cost.
Efficiency and Safety
Many of us are forced to undergo invasive surgery at some point in our lives, followed by a recuperation period proportional to the seriousness of the procedure. Older patients are more likely to undergo invasive medical procedures, and their recovery process is longer. The Medigus camera, which is affixed to special devices, is minimally invasive and eliminates the arduous recovery process from surgery. Medigus’s SRS endoscope can substitute standard surgery for gasrto-esophageal reflux disease (GERD), caused by stomach fluid rising into the esophagus. GERD is one of the most common diseases plaguing the Western world, afflicting an estimated 7% of the population.
The Medigus micro-devices can also efficiently diagnose gall stones, which are currently diagnosed with X-rays or expensive and fragile optical fibers. Medigus’s micro-camera spares the damages of X-ray radiation as well as the costliness of optical fibers.
The Medigus camera has enabled doctors, for the first time in history, a noninvasive glance into the heart, brain and other areas. Dr. Sonnenschein estimates that within 5-10 years, the technology will be used in dozens of procedures including vascular treatments, from inserting stents to replacing valves.
According to Dr. Sonnenschein, the camera technology could be utilized to effectively substitute invasive procedures carried out in hospitals, especially in the fields of orthopedics and ear-nosethroat medicine with less invasive procedures carried out at private clinics and outpatient clinics, offering a more efficient and cost effective option.
Abstract
Recent medical trends have highlighted the functionality and value of miniature cameras. The quality of healthcare can be significantly enhanced by affording direct vision of otherwise inaccessible organs. Miniature cameras, commonly denoting a total diameter of less than 3mm to 3.2mm, present a new set of challenges together with their inherent benefits.
This paper reviews the main shifts in medical practice and equipment that paved the way for the acceptance of miniature cameras, followed by a discussion of the main elements of such changes and an illustration of the benefits of direct vision in several select applications.
1. Introduction
Of the prevalent trends in medicine during the last two decades, some are purely science-driven, a few are rooted in patient attitudes and others are influenced by the advancement of modern technology. To name a few of these trends, medicine is becoming more and more evidence-based, whereby physicians are less inclined to rely on subjective interpretations and instead seek documented measurements using sensors and imaging devices, as well as population surveys and biological indicators.
Conversely, current medicine is drifting towards a personal orientation, not in terms of physicians’ personal attitudes, but in the sense that each patient represents a unique biological system that shares many general characteristics with the human species, while concurrently constituting a unique manifestation. As a phenotype, each patient warrants personally-tailored therapy, protocols and medical treatment routines , from diagnosis through follow-up. Thus, scientific justification of modern medicine is gained through the careful analysis of the population at large, but treatments are derived and formulated via careful consideration of the individual patient.
Developments in information technology facilitate the accumulation and accessibility of data that can be analyzed at a later date in order to gain insights with regards to maladies, pathologies and curing methods. Moreover, when combined with the consistently increasing utilization of information sources and improved analysis techniques, this surge of information will frequently provide convincing evidence that may be used to refute long-standing medical misconceptions, thereby improving patient management.
The information explosion prompts new modalities to promote better understanding of the causes and evolution of illness, with imaging modalities occupying a dominant position. Better imaging, as a term, encompasses quite a few notions: more realistic imaging such as three-dimensional stereoscopic imaging, higher resolution, images from previously unattainable locations or angles of view, and different wavelength ranges facilitating otherwise undetectable features of the human body. Good examples of the latter are X-ray, gamma-ray (PET), infrared, radio-frequency and more.
Different wave-forms, like ultrasound, have also been helpful where visual, electro-magnetic techniques fail to obtain the necessary data. Parallel to the widening of the frequency and wave-form horizons, two other critical abilities have emerged: the combination of many still images or video streams into a single multi-faceted view of the human body, e.g. computerized tomography (CT), and the increasing computational power that has been harnessed to augment imaging information through image processing (see e.g., (Rabbetts 2007)).
The concept that computer based processing of accumulated digital “raw” data could portray a clearer view of the object’s condition led to computer-aided-diagnosis or computer-aided-technology (CAD/CAT) aiming to help physicians achieve more accurate diagnoses, higher sensitivity levels and somewhat better specificity in many routine tasks (Henning Müller 2004). Examples range from nodule or polyp identifications within documented scans to automatic cell counting under the pathologist’s microscope.
The multiple modalities that are now available have also contributed to the use of multiple viewing angles that may allow physicians to better discern the patient’s condition. As an example, registrations of MRI and CT scans have resolved the shortcomings of each separate modality.
The introduction of the ‘personalized medicine’ notion and the availability of imaging modalities with computerized analysis soon led to accurate localization of pathological conditions (Bankman 2008). This localization, in turn, facilitated pinpoint treatment such as interventional chirurgical procedure. High-precision abilities have enabled these procedures to be conducted in a minimally invasive and well localized manner. When invasive treatment is avoided as much as possible, the general well-being of the patient increases.
It has become clear that patient comfort and well-being, as trivial as this may sound, contributes tremendously to the success of the treatment. Thus, minimally invasive methods emerged as the state-of-the-art in patient diagnosis and treatment. When invasiveness cannot be altogether avoided, it is only gradually introduced.
Attempts are made to first utilize natural orifices, either as a direct portal to the region of interest or as an enabling passage to the final destination using miniature incisions. Thus, of the notion that a single incision (and a small one at that!) should be regarded as a port for laparoscopic procedures or NOTES (Natural orifice Translumenal Endoscopic Surgery (Chamberlain RS 2009, Abu Gazala 2012)), for example, brought a significant advantage to the standard of care for many conditions that otherwise would have been treated with the vigorous use of a scalpel.
These advancements acted as a call for technology development. The ability to refrain from making incisions in order to view internal organs, or alternatively, to rely on smaller and smaller incisions and operating tools, suggested that vision, either direct or indirect, may also be useful and necessary in order to take full advantage of these minimally-invasive techniques (Rosen M 2011).
Continuous view (i.e., video) lends another aspect to the treatment – the capability to “close the loop”, namely to monitor the diagnosis (e.g., biopsy) or treatment in real time, change the tools, adapt the maneuvers, and improve the protocol according to the visual information, while directly observing the effect of these changes.
It is precisely at this point that miniature cameras enter the picture. The combination of evidence-based (i.e., image) medicine, with the consideration of the patient’s individual anatomy and condition (“personalized medicine”) together with the newly-gained information and the extraction of the valuable parameters from it, demands the use of miniature video cameras. When technology enabled low-cost manufacturing, the disposable nature of the cameras has also played an important role in their desirability for use in medical devices which are more commonly intended for single use.
Reservation of the term “miniature camera” for outer diameters of less than 3mm to 3.2mm, as specified earlier, is mainly derived from the inner diameter of standard endoscope working channels. However, today’s cameras may be far smaller than these dimensions and are not necessarily introduced through an endoscope’s working channel.
In summary, recent trends in medicine support each other and spur yet more advancement that in turn provokes science and technology to devise new methods and to improve upon existing technology to catch up with the exacting requirements of modern medicine.
2. Imaging modalities – limitations and challenges
As much as medicine attempts to be an advanced and cutting-edge quasi-science, it is still, to the benefit of us all, a rather conservative field. The progress in medicine and the paradigm shifts occur at a relatively slow pace, both for the sake of patient safety and due to economic factors.
Since the biggest body of evidence to date for the diagnosis of many pathologies is based on visual inspection by the human eye (colors, shapes, sizes, etc.), whether in-vivo or in-vitro, physicians prefer to continue relying on such routines when they diagnose and evaluate pathological conditions.
In the frequency spectrum visible to the unaided eye, and in abutting frequencies (near infrared and ultraviolet), it is common to assess and evaluate the tissue or organ condition through visual inspection and its technological counterpart – photography. The fact that photography (or filmography) does not exactly manifest the same impression as the one obtained by the naked eye should be taken into account, but should not be a deterring factor for the use of the former as a diagnostic means.
The twentieth century semi-conductor technology produced the CMOS sensors as affordable – and more importantly, accurate and scalable – devices for visual imaging needs. Medicine soon adopted these solutions for its own needs, which paved the road to visual imaging in many common clinical scenarios. The marriage between fiber-optics technology and visual imagers allowed, for the first time, in-vivo visual exploration inside the human body, without the need for highly invasive surgery.
However, the motivation for miniature cameras goes beyond the mere ability to penetrate narrow, small locations within the human body. The benefits of smaller incisions are clear: lower risk, faster healing and recovery, smaller chance for related infections, reduced sedation and pain and aesthetic appearance (scars). All of these may lead to an additional advantage which can be superficially seen as purely economic – the transition of procedures from the operating room to office-based clinics.
Better yet are the zero-incision procedures that have become feasible by the introduction of miniature cameras. Trans-oral procedures that may involve intubation can now be performed transnasally, even in children. No harm is caused by the examination, and this alone may cause the procedures to be so appealing as to be considered screening routines rather than symptom-triggered tests.
Going beyond visualization per-se, the integration of mini-cameras within endoscopes of all sorts vacates precious space for other elements within the endoscope and allows more complicated procedures to be performed via surgical endoscopy. A useful embodiment of such utilization is found in the “mother – baby” endoscope combination, in which a small-diameter camera system is threaded through the working channel of the “mother” scope.
This arrangement enables the removal of the visualization tool and the use of the channel for a different tool as required during the endoscopic session. The “mother” endoscope serves as a robust, fully steerable modality supporting the somewhat less controllable baby-scope. The baby-scope can nonetheless reach places that are beyond the reach of the mother scope due to its smaller size and lack of certain physical and anatomical constraints. One notable usage of this method can be found in the NOTES-type procedures that accesses the peritoneum through the stomach.
3. Visualization methods: Fibers Versus Cameras
3.1 Fiber-based cameras
Upon the introduction of miniature cameras, the natural tendency would have been to combine them with optic fiber technology. Optic fiber bundles containing thousands of individual fibers, each one acting as a mirroring channel, may be connected directly to the cameras. Optics are usually placed at the distal tip of the bundle, on top of its polished edge. Nevertheless, the distal tip optics tend to be one of the vulnerabilities of this technology. At the bundle proximal end, there is usually a coupler to the camera to facilitate the best image quality possible.
This arrangement entails several shortcomings. First, the common fibers have a circular cross section which causes “blank spaces” between the bundled fibers. Although hexagonal shape fibers have been proposed, their use is not widespread due inter alia to high costs and complicated manufacturing. This brings about another limitation of the fiber solution, namely the cost per performance factor.
Usually, vision optic fibers, as opposed to illumination fibers, are rather expensive. Had they been robust, they could have been a cost-effective solution. Unfortunately, the fragility of the fibers prevents them from being a truly multiple-use device, especially if they are not placed in a rigid endoscope.
The flexible endoscopes/catheters pose an acute challenge for fiber-based devices. The limited curvature radius that the bundles can withstand without breaking is a severe constraint when it comes to small cameras whose main advantage is their ability to accommodate narrow, winding lumens.
Sharp turns, even when generally within device specifications, end up causing fractures along several or even many of the fibers. Such fractures result in immediate image quality deterioration.
Another obstacle that may arise during the design and implementation of fiber-based medical devices is the somewhat limited field of view.
Since fiber-based solutions necessarily dictate at least two coupling elements (at the tip and then at the proximal end), the tip coupling suffers from the optical matching of the lens (if any) with the polished fibers behind it. This usually restricts the field of view (namely the opening angle) of the front lens and thus constrains the device’s function. The rear (proximal) end coupling must be performed in a very precise way in order to bring the bundle plane to the focal plane of the proximal camera, when the two planes are close to each other for full coverage of the bundle.
Another potential shortcoming of fiber-based devices may be expressed when the desired angle of view is different than 0º. There are two alternatives to meeting this requirement: the fiber tip maybe polished diagonally to accommodate the desired angle and acts like a periscope mirror or a prism, or else, the prism is coupled directly to the tip. Both solutions carry unwanted consequences: the diagonal polish limits the use within a specific medium (e.g. water) and precludes a full (round) image when introduced to a different medium (e.g. air). The prism solution makes the abovementioned coupling between the distal tip and the prism even more challenging and casts an additional cost burden on the device. Fiber-based scopes may go to diameters of ~1mm.
Most of the abovementioned limitations of fiber-based imaging solutions are alleviated by the use of a distal tip camera, instead of at the proximal end where the light must first pass through the fiber bundle. When the camera is situated at the front end of the device, it is the element that first encounters the collected light. Thereafter, an electric wire transfers the video signals detected by the camera.
3.2 Camera at tip solution
Even though the camera at tip solution is usually superior to the fiber-based solution, it bears its own challenges. Most of these are directly derived by the camera dimensions that dictate unconventional solutions for packing, processing and control of the camera.
The basic element of all cameras is the sensor that is situated behind the lens, its main characteristics being pixel size and pixel density (namely the number of pixels and the size of the sensor). For some applications the sensor sensitivity and its dynamic range are also of concern.
In this solution, the optics of the camera is the element which should satisfy the field of view and the angle of view (the main optical axis direction with respect to the endoscope main axis) as deemed by the application.
The optics and the sensor are embedded together within the “house” or the “barrel”, with or without adjusting the optic-sensor distance to provide the desired focus, focal depth, field of view, etc.
The sensor can be wired by various techniques, but usually direct placement onto a PCB or wire-bonding is used. In the production process, special attention is paid to accurate alignment between the optics, the sensor and its placement. This not only contributes to improved image quality, but also enables the minimization of the camera’s overall dimension.
The electric wires cannot exceed the camera diameter, without defeating the raison d’être of the miniature camera. This requirement is not a trivial one, due to the requirement for shielding to reduce electro-magnetic interference.
Custom filters, coating materials, connectors, etc. are also usually present within each camera system and should be coordinated with the sensor micro-lenses, Bayer format as well as outer parameters such as the expected illumination, environment and more.
In the camera in tip solution, the angle of view can be changed by a prism, like in the fiber-based case, with a prism-lens perfect coupler. Better yet, due the small length dimension of these cameras, some of them can be placed at an angle to the main mechanical axis of the endoscope, thereby resolving this issue altogether. Camera at scopes tip may go today down to outer diameters of ~1mm (e.g., ScoutCam).
The camera does not transmit a visible image, but is connected to an image processing unit that must also be synchronized and built according to the camera specifications.
In short (no pun intended), the main challenges in miniature camera implementation and production are (i) Small dimensions, not only the overall diameter but also the length dimension – a long rigid camera is not practical when inserted into a narrow meandering lumen; (ii) Accurate assembly and – in particular – precise optical alignment; (iii) Application-oriented design and implementation; (iv) Full match between the camera specifications and the accompanying image processing equipment; (v) Tight linkage between the camera assembly and its surroundings (e.g. illumination) (vi) Cost-effective assembly.
Keep in mind that being a medical camera, all of the camera elements to which the patient may be exposed must be strictly bio-compatible. Disinfection or sterilization in-between patients can be avoided altogether by the virtue of the single-use, cheap camera.
In section 4 we will refer to different degrees of bio-compatibility with regards to the applications that the camera should serve.
3.3 Other solutions
One type celebrated miniature camera is embedded within medical capsules (“pill-cams”). Three main differences exist between the video cameras discussed here and the pill cameras. First, the size of the pill camera does not strictly fit in the “miniature camera” category. The second difference is the pill’s independence in terms of its motion and power. This independence is technically restrictive (battery lifetime, strict power management, inability to steer or maneuver) but clinically advantageous for specific applications.
For instance, the patient’s freedom while under pill inspection cycle and the prolonged duration of the inspection are two positive aspects of the pill camera independence. The third difference is the fact that the pill does not have to show real time images. When the pill travels at a slow enough pace, its power management calls for a slowdown of the frame rate without an increase in latency due to the “nonreal-time” mode of operation. Needless to mention is the fact that the pill is also disposable.
4. The opportunity with small cameras
One of the main areas in which miniature cameras can demonstrate a real advantage is in the field of modern medicine. As described in section 1, modern medicine is becoming increasingly inclined toward minimally invasive procedures, whether diagnostic tasks or true treatments.
Miniature cameras provide a natural element within this trend, with which the physicians can gain valuable, direct information that formerly was only possible to obtain after surgical openings or frequencies outside the visible spectrum (X-ray, infrared, etc.).
We have named several fields of medicine in which miniature cameras may, and in some cases already do, provide indications and data to improve healthcare. Lumens of all sorts are the obvious candidate for miniature camera diagnoses. In the cardiovascular field, the main challenge is to clear out blood in order to enable visual inspection of blood vessels, for example. Other lumens are less problematic (e.g. larynx, pharynx, esophagus, ear canal, nostrils) but some are collapsed in their regular condition and must be manipulated in order to exploit the full view with the miniature camera aid.
In natural cavities, one of the main challenges is to provide sufficient illumination (if the cavity is spacious and a global view is required). For large cavities, the finite dynamic range of the cameras also complicates simultaneous viewing of nearby and more distant objects. However, even for a large dynamic range, a real time application usually calls for minimal latency and therefore limits the maximum allowed exposure time to compensate for poor illumination.
This is a major difference between still images of all sorts, or semi-still (namely low video rate, see above discussion of the “pill camera”) and a high video rate camera. The current standard rate is 30 fps (frames per second).
One of the most complicated tasks is the a-priori simulation or design of the illumination conditions prevalent within a cavity or a lumen. The unknown structure, reflections, tissue condition and so on, prevent the designers from applying a single solution to multiple applications. As a result, the development process of a device for specific applications is necessarily an iterative process that should encompass all aspects of the expected environment.
Single incision applications may also serve as an appropriate platform for miniature cameras, with or without robotic technology (Oleynikov D 2005, Ahmed I 2011, Otten ND 2011, Greaves N 2011). Such procedures can replicate laparoscopic procedures with smaller incisions and trocars, to bring the latter closer to the object under inspection and to circumvent the inconvenience and limitations of rigid laparoscopes. This is particularly relevant in view of the smaller diameter of the miniature camera that makes room for more steering mechanisms.
Rigid endoscopes enjoy the same benefits and are usually needed in one of two occasions: when lateral force or moment is applied in order to adjust the direction of view so that it meets the region of interest (e.g. arthroscopy cameras) or when the camera itself acts as a needle to penetrate through a wall.
In general, the flexibility of the miniature camera enables it to wind around obstacles and within lumens, and to penetrate strictures and walls if used in the context of rigid endoscopes. This is also evident when used in combination with the mother-endoscope vehicle. The mother-endoscope provides the gross steering, the capability to apply force on the lumen or tissues and acts as a protective shield for the more delicate mini-camera. The mini-camera goes the “last mile” and peeks at more distant objects where the mother-scope dimensions do not allow a close look. It can also use the mother outlet structure as a leaning wall or even a direction changer, like in ERCP procedures (Chamberlain RS 2009).
The specific application is also what dictates the level of disinfection or sterilization the miniature camera needs to withstand (a single time, during production). This may range from simple disinfection for truly non-invasive applications (e.g. ear canal) all the way to high sterilization degree when introduced into a blood vessel.
Clearly, the advantages of the small dimensions of such cameras are fully exploited in combination with other tools such as etchers, biopsy collectors, object removers, etc. A separate, dedicated working channel for the vision device or a common channel that accommodates other tools according to the immediate needs are both possible.
Recently, a few innovative augmenting elements have been introduced to make yet better use of miniature cameras. Some are mature and ready for integration with miniature camera technology, while others are at a more preliminary stage, but nonetheless hold significant promise for new types of treatment. These elements include a miniature integrated zoom mechanism, an electric steering mechanism, magnetic steering capabilities, stereoscopic view as derived by multiple camera utilization, and more.
5. Summary and Conclusions
Miniature cameras can be merely referred to as gadgets, however a closer look at the enabling technology they introduce, reveal their real benefits. These cameras are already changing the way physicians are gathering the necessary information in order to reach at correct patient management decisions. The ability to monitor procedures, simple or complicated, a larynx inspection or ERCP stone removal, modifies the way these procedures are conducted.
The demand for sophisticated medical interventions and technological progress are two elements in a closed loop that feed each other. Even though miniature cameras are not yet as good, in terms of image quality, as big, amateur cameras, they have become indispensable. The need to see inner organs through small windows or through orifices together with the desire to monitor therapies are by now part of the standard care for some conditions. We foresee that in the near future, it would consider mal practice when a physician will not be using the option to judge by direct vision and to treat with eyes wide open. Every organ that is accessible will be accessed – big and small. The miniature cameras will serve this function to provide better, more precise patient care management.
6. Acknowledgements
Ts.K. would like to thank Aaron Jaffe for his valuable comments on the manuscript.
7. References
Abu Gazala, Shussman, Abu Gazala, Schlager, Elazary, Ponomernco, Khalaila, Rivkind, and Mintz. “Miniature Camera for Enhanced Visualization for Single-port Surgery and Notes.” Journal of Laparoendoscopic & Advanced Surgical Techniques , 2012: 22 (10) 984-8.
Ahmed I, Paraskeva P. “A clinical review of single-incision laparoscopic surgery.” The Surgeon : Journal of the Royal Colleges of Surgeons of Edinburgh and Ireland, 2011: 9(6):341-351.
Bankman, I. Handbook of Medical Imaging. Processing and Analysis. Burlington: Academic Press, 2008.
Chamberlain RS, Sakpal SV. “A comprehensive review of single-incision laparoscopic surgery (SILS) and natural orifice transluminal endoscopic surgery (NOTES) techniques for cholecystectomy.” Journal of Gastrointestinal Surgery : Official Journal of the Society for Surgery of the Alimentary Tract , 2009: 13(9):1733-1740.
Greaves N, Nicholson J. “Single incision laparoscopic surgery in general surgery: a review.” Annals of the Royal College of Surgeons of England, 2011: 93(6):437-440.
Henning Müller, Nicolas Michoux, David Bandon, Antoine Geissbuhler. “A review of content-based image retrieval.” International Journal of Medical Informatics, 2004: (73) 1-23.
Oleynikov D, Rentschler ME, Dumpert J, Platt SR, Farritor SM. “In vivo robotic laparoscopy.” Surgical Innovation, 2005: 12 (2) 177-181.
Otten ND, Farritor SM, Lehman AC, Wortman TD, McCormick RL, Markvicka E, Oleynikov D. “Miniature in vivo cameras for use in single-incision robotic surgery – biomed 2011.” Biomedical Sciences Instrumentation, 2011: 47:165-170.
P. Coan, A. Peterzol, S. Fiedler, C. Ponchut, J. C. Labiche and A. Bravin. “Evaluation of imaging performance of a taper optics CCD `FReLoN’ camera designed for medical imaging.” J. Synchrotron Rad., 2006: (13) 260-270.
Rabbetts, Ronald B. Clinical Visual Optics. London: Elsevier, 2007.
Rosen M, Ponsky J. “Minimally invasive surgery.” Endoscopy, 2011: 33(4):358-366.
Simi M, Pickens R, Menciassi A, Herrell SD, Valdastri P. “Fine Tilt Tuning of a Laparoscopic Camera by Local Magnetic Actuation: Two-Port Nephrectomy Experience on Human Cadavers.” Surg Innov., 2012.
COVID-19 had a particular impact on medical processes, with the effect on the medical device industry being twofold. On the one hand, the supply chains of all companies, from start-ups in their first years to global leaders, were severely disrupted.
Many sub-tier suppliers stopped production or at least drastically slowed it under the strict isolation restrictions. Traveling restrictions negatively affected the interaction between engineers and quality teams from both sub-suppliers and device manufacturers.
On the other hand, except for some specific product lines, demand for devices dropped in light of the pause on elective surgeries.
Emerging Trends
As the medical supply chain was forced to shift during the pandemic, interesting trends emerged. First, medical teams became society’s heroes while public attention was focused on their challenging and demanding work, especially with the healthcare supply chain disrupted.
Second, tremendous financial and medical resources were reallocated to meet demand for the tools needed to combat COVID-19, such as ventilators. Other, less relevant devices were a lower priority.
Lastly, remote communication, mainly based on video, became the hallmark of this era. Reliance on video as a major source of communication is here to stay.ScoutCam integrated solutions
The use of video technology allows for remote diagnostics, presents an incisionless way to get information from inside the body and permits the use of minimally invasive therapeutic procedures. Hence, video technology, and the production and delivery of these products, plays a critical role in a time when only essential surgeries are taking place.
Beyond video, the innovation that will play a larger role in the medical world should we face another disruption in our daily lives is medical robotics (which, in turn, is directly related to video technology). In addition, the best practices established under strict limitation and constraints might have long-term, positive impacts on the industry.
Yaron Silberman is CEO of ScoutCam, which develops and manufactures customized visualization solutions.